ISO 14971 Explained – Risk Management in Medical Devices
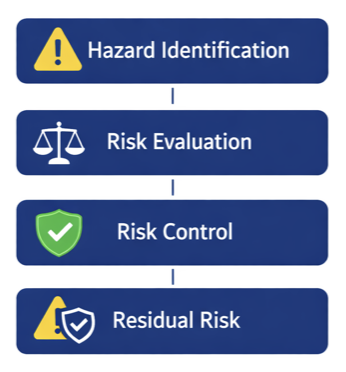
ISO 14971 Explained – Risk Management in Medical Devices Risk management is one of the most critical aspects of medical device development. Every device, from simple tools to complex software-driven systems, must be safe for patients and users. ISO 14971 defines the structured process used to identify hazards, analyze risks, and implement effective control measures …
ISO 14971 Explained – Risk Management in Medical Devices Read More »