Medical Device Standards Explained
How ISO 14971, IEC 62304 & ISO 13485 Work Together
Medical device development is one of the most highly regulated engineering domains. Unlike many other industries, safety, quality, and software development are governed by strict standards that must work together seamlessly.
Three of the most important standards are:
- ISO 14971 – Risk Management
- IEC 62304 – Software Lifecycle
- ISO 13485 – Quality Management System (QMS)
Understanding how these standards interact is essential for engineers, quality managers, and regulatory professionals.
In this article, we break down each standard and explain how they work together in real-world medical device development.
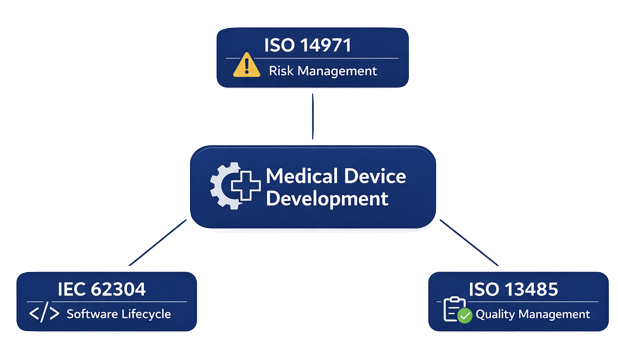
Why Multiple Standards?
At first glance, having multiple standards may seem unnecessary. Why not just one?
The reason lies in complexity and specialization.
Medical devices combine:
- Hardware systems
- Embedded software
- Risk-critical functionality
- Regulatory requirements
Each standard focuses on a specific aspect:
- Risk → ISO 14971
- Software → IEC 62304
- Organization & processes → ISO 13485
Together, they form a complete framework for safe and compliant product development.
The Three Key Standards
To understand the big picture, it’s helpful to see these standards as complementary layers:
- ISO 14971 ensures risks are identified and controlled
- IEC 62304 ensures software is developed safely
- ISO 13485 ensures the entire organization follows structured processes
They are not independent—they are deeply interconnected.
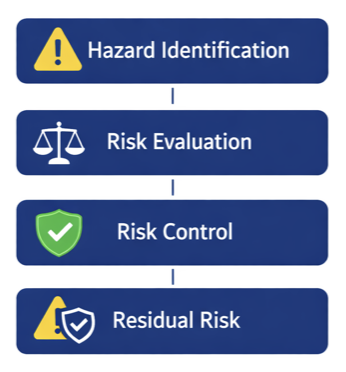
ISO 14971 – Risk Management
ISO 14971 defines how risks are managed throughout the lifecycle of a medical device.
Key concepts:
- Hazard identification
- Risk analysis and evaluation
- Risk control measures
- Residual risk assessment
- Post-market surveillance
Risk management is not a one-time activity—it is continuous.
Every design decision, software function, and system interaction must be evaluated in terms of risk.
Example:
A software malfunction could lead to incorrect patient data → this is identified as a hazard → risk is assessed → mitigation is implemented.
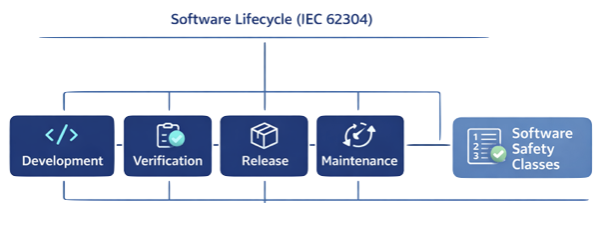
IEC 62304 – Software Lifecycle
IEC 62304 focuses on software development processes for medical devices.
It defines:
- Software safety classification (Class A, B, C)
- Development lifecycle processes
- Verification and validation
- Maintenance and updates
- Problem resolution
Software is one of the biggest risk drivers in modern medical devices.
That’s why IEC 62304 is tightly linked to ISO 14971:
Software risks identified in ISO 14971 must be addressed through IEC 62304 processes.
ISO 13485 – Quality Management System
ISO 13485 defines how organizations manage processes to ensure consistent quality and compliance.
It includes:
- Process management
- Documentation and traceability
- Management responsibility
- Supplier control
- Audits and CAPA (Corrective Actions)
ISO 13485 ensures that:
Risk management (ISO 14971) and
Software development (IEC 62304)
are not performed in isolation, but within a controlled and auditable system.
How They Work Together
The real value comes from integration.
Example workflow:
- A hazard is identified → ISO 14971
- A software-related risk is derived → ISO 14971
- Software is developed to mitigate it → IEC 62304
- The process is documented and controlled → ISO 13485
This creates a closed-loop system:
- Risks drive development
- Development mitigates risks
- Processes ensure consistency and compliance
If you want to learn how to apply Medical Device standards step by step in real projects:
Comparison to Other Industries
Medical device development differs significantly from other industries like automotive or aerospace.
Key differences:
- Stronger regulatory focus (FDA, EU MDR)
- Higher documentation requirements
- Strict audit expectations
- Lifecycle-based compliance
While automotive uses standards like ISO 26262, medical focuses more on:
Risk + Compliance + Documentation
This makes understanding these standards even more critical.
Challenges in Practice
In real projects, companies often struggle with:
- Integrating multiple standards
- Aligning risk management with software development
- Maintaining documentation consistency
- Preparing for audits
- Handling changes and updates
One common issue is treating standards as separate silos instead of an integrated system.
Common Misunderstandings
Some typical misconceptions include:
❌ “ISO 14971 is only documentation”
→ It is a core engineering activity
❌ “IEC 62304 is just for software teams”
→ It impacts system design and risk handling
❌ “ISO 13485 is only for quality departments”
→ It governs the entire organization
❌ “You can apply standards independently”
→ They must be integrated
Understanding these misunderstandings is key to avoiding costly mistakes.
Summary
Medical device development relies on a combination of standards that work together:
- ISO 14971 → manages risk
- IEC 62304 → structures software development
- ISO 13485 → ensures organizational compliance
Together, they create a complete framework for building safe, reliable, and compliant medical devices.
If you understand how these standards interact, you gain a major advantage in engineering, quality management, and regulatory work.



