ISO 14971 Explained – Risk Management in Medical Devices
Risk management is one of the most critical aspects of medical device development. Every device, from simple tools to complex software-driven systems, must be safe for patients and users.
ISO 14971 defines the structured process used to identify hazards, analyze risks, and implement effective control measures throughout the entire lifecycle of a medical device.
In this article, you will learn how risk management works in practice and why it is essential for regulatory compliance.
Why Risk Management Matters
Medical devices operate in safety-critical environments. Even small failures can lead to serious consequences.
Risk management ensures that:
- Potential hazards are identified early
- Risks are systematically analyzed
- Appropriate control measures are implemented
- Devices remain safe throughout their lifecycle
Without a structured risk management process, it would be impossible to ensure compliance with regulations such as FDA requirements or EU MDR.
Risk management is not optional – it is a core requirement.
What is ISO 14971?
ISO 14971 is the international standard for risk management of medical devices.
It defines:
- A systematic risk management process
- Responsibilities and documentation requirements
- Continuous lifecycle integration
- Post-production monitoring
The standard is not just about documentation—it is about engineering decisions based on risk.
It ensures that safety is built into the product from the very beginning.
Risk Management Process
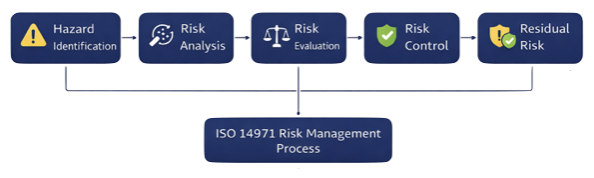
The ISO 14971 process follows a structured sequence:
1. Hazard Identification
Identify potential sources of harm, such as:
- Software errors
- Hardware failures
- User misuse
2. Risk Analysis
Estimate the risk by evaluating:
- Severity of harm
- Probability of occurrence
3. Risk Evaluation
Compare the estimated risk against predefined acceptance criteria.
Decide whether the risk is acceptable or requires mitigation.
4. Risk Control
Implement measures to reduce risk:
- Design changes
- Protective mechanisms
- Information for safety (e.g. warnings)
5. Residual Risk Evaluation
After applying controls:
- Assess remaining risks
- Perform benefit-risk analysis if necessary
6. Post-Production Activities
Monitor the device after release:
- Collect field data
- Identify new risks
- Improve the product continuously
This process is iterative and continues throughout the entire lifecycle.
Outputs of Risk Management
ISO 14971 requires clear documentation and traceability.
Key outputs include:
- Risk management plan
- Risk analysis documentation
- Risk control measures
- Residual risk evaluation
- Risk management report
These outputs are essential for:
- Regulatory approval
- Audits
- Internal quality assurance
If you want to learn how to apply ISO 14971 step by step in real projects:
Role within Medical Device Development
Risk management is not isolated—it is integrated into the entire development process.
It directly interacts with:
- IEC 62304 (Software)
→ Software risks must be identified and controlled - ISO 13485 (QMS)
→ Risk management must be documented and controlled
Example:
A software function is identified as safety-critical →
Risk is analyzed (ISO 14971) →
Software is developed with controls (IEC 62304) →
Process is documented (ISO 13485)
Risk management drives engineering decisions across the system.
Challenges in Practice
In real-world projects, companies often face challenges such as:
- Identifying all relevant hazards
- Defining realistic risk acceptance criteria
- Ensuring consistency across teams
- Maintaining traceability between risks and controls
- Integrating risk management into development workflows
A common issue is treating risk management as a documentation exercise instead of an engineering activity.
Common Misunderstandings
Several misconceptions can lead to serious issues:
❌ “Risk management is only paperwork”
→ It directly influences system design
❌ “Risk analysis is done once”
→ It must be continuously updated
❌ “Only safety engineers are responsible”
→ It involves the entire development team
❌ “Residual risk can be ignored”
→ It must always be evaluated
Understanding these points is critical for successful implementation.
Summary
ISO 14971 defines how risks are managed in medical device development.
- It provides a structured and continuous process
- It ensures safety is integrated into design
- It supports regulatory compliance
- It connects engineering, software, and quality processes
Risk management is not just a requirement—it is a core engineering discipline in medical device development.



